 이미지 확대보기
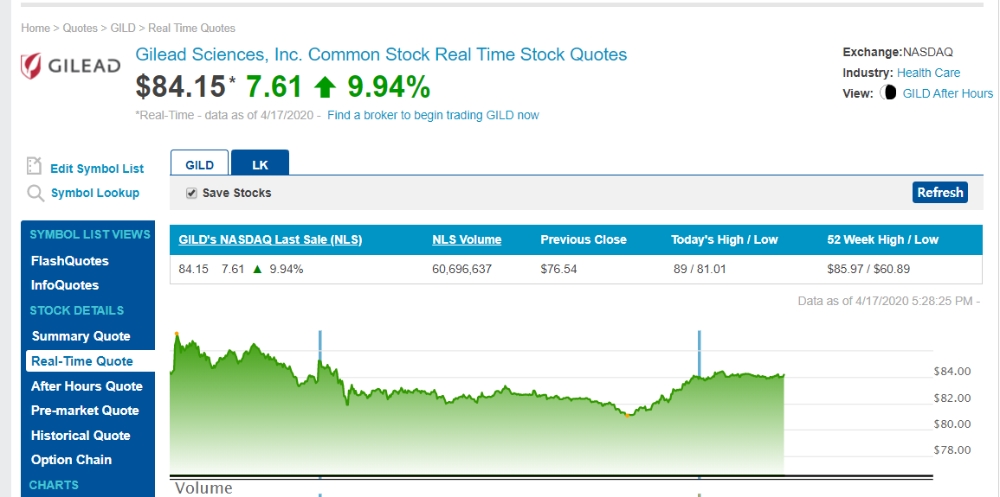
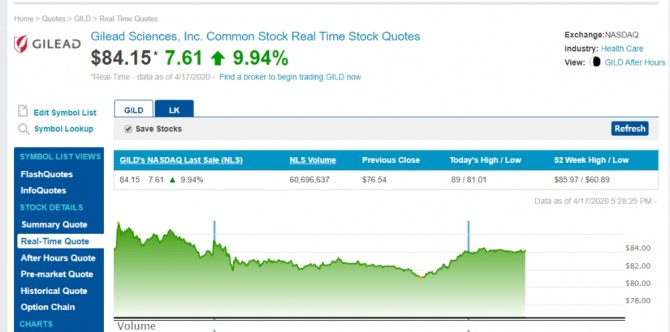
이미지 확대보기렘데시비르 관련주 길리어드(Gilead)의 주가 변동내역
Best Bid / Ask$ 83.85 / $ 83.99
Today's High / Low$ 85.79 / $ 81.01
Share Volume94,044,087
50 Day Avg. Daily Volume23,136,580
Previous Close$ 76.54
52 Week High / Low$ 85.97 / $ 60.89
Market Cap105,744,684,128
P/E Ratio19.95
Forward P/E (1y)12.74
Earnings Per Share (EPS)$ 4.21
Annualized Dividend$ 2.72
Ex Dividend Date3/12/2020
Current Yield3.64 %
Beta 1
뉴욕증시 에서는 렘데시비르 관련주 폭등을 몰고온 길리어드(Gilead)란 회사에 관심을 집중하고 있다.
코로나 치료제 정말 나오나 는 기대와 의문이 교차하면서 미국 FDA의 공식 평가를 기다리고 있다.
18일 뉴욕증시에 따르면 미국의 의료 전문 매체인 STAT뉴스는 시카고대 연구진이 환자들에게 렘데시비르를 투약한 결과 대다수가 발열 및 호흡기 증상이 빠르게 회복돼 1주일도 안되어 퇴원할 수 있었다고 보도했다. 이에대해 길리어드는 아직 임상 시험 최종 결과가 나온 것은 아니라면서 신중한 입장이다.
길리어드는 조만간 코로나19 중증 환자에 대한 렘데시비르 임상시험 결과를 공식 발표할 예정이다.
미국의 경제 재개 움직임이 가시화된 것도 미국 뉴욕증시에 호재가 됐다. 트럼프 미국 대통령은 전일 '미국의 재개'라고 명명한 3단계 경제 활동 정상화 지침을 발표했다.
길리어드 사이언스의 주가는 이틀 연속 폭등했다. 렘데시비르 관련주의 단초는 미국 메디컬 매체인 스탯 뉴스가 제공했다. 미국 메디컬 매체인 스탯 뉴스는 시카고대 3단계 임상실험 에서 코로나19로 입원한 환자들 렘데시비르 치료 이후 열과 호흡기 증상이 크게 완화되었으며 1주일내에 퇴원했다고 보도했다. 이 보도가 나온후 뉴욕증시 나스닥에서는 길리어드 사이언스의 주가가 폭등했다. 한국 코스닥 에서도 렘데시비르 관련주로 뷴류되는 파미셀 한올바이오파마, 엑세스바이오,에이프로젠제약,신풍제약, 진원생명과학등의 주가가 크게 변동했다.
다음은 렘데시비르 관련주, 길리어드 사이언스의 홈페이지 안내문
1987년에 창립된 길리어드 사이언스는 올해로 창립 30주년을 맞았습니다. 미국 캘리포니아의 작은 벤처기업으로 시작하여 2016년 현재 글로벌 총 매출액 326억 달러(약 38조원), 시가총액 1,231억 달러(약 144조원), 글로벌 9위 규모의 글로벌 톱 클래스 제약 기업으로 빠르게 성장하였습니다. 길리어드는 의약적 요구가 충족되지 않은 분야에서 혁신적 치료제 개발에 집중해 오고 있으며 특히 항바이러스 치료제 분야에서 독보적인 역량을 보유하고 있습니다. 그 동안 계속된 치료제의 개발로 HIV를 불치의 병에서 만성질환으로 치료의 페러다임을 바꾸었고 B형간염을 위해 약물 치료를 받아야 하는 환자들의 삶의 질을 현격하게 개선하였으며 C형 간염의 경우 ‘치료가 힘든 질환’을 ‘완치’가 가능하게 하는 혁신을 이루어 내었습니다.
Gilead Sciences,
An Open Letter from our Chairman & CEO
Daniel O’Day - April 10, 2020
Earlier today, the New England Journal of Medicine (NEJM) published an analysis of the effects of our investigational medicine remdesivir on a small group of patients with severe symptoms of COVID-19.
These are patients who received treatment through the compassionate use program for remdesivir, which is for critically ill patients who are unable to take part in a clinical trial. The results, which cover 53 of the first patients to have been treated in the program, show that the majority demonstrated clinical improvement after taking remdesivir. We recognize the limitations of these compassionate use data from a purely investigational perspective, while knowing they are of the greatest significance for the patients whose symptoms improved. These early data from 53 patients have not been generated in a clinical trial and cover only a small portion of the critically ill patients who have been treated with remdesivir.
Remdesivir is an investigational treatment and has not been approved for use anywhere in the world. In the broader efforts to determine whether it is a safe and effective treatment, we have some way to go. Multiple clinical trials are underway across the world to build a complete picture of how remdesivir works in various contexts. These studies cover a range of patient populations across different demographics and with varying types of symptoms: moderate, severe where patients need oxygen support, and critical where medical ventilation is required. These patients all receive remdesivir through intravenous infusions in a hospital setting.
In studying remdesivir, the question is not just whether it is safe and effective against COVID-19 but in which patients it shows activity, how long should they receive treatment and at what stage of their disease would treatment be most beneficial. Many answers are needed, which is why we need multiple types of studies involving many types of patients.
Some of these answers will start to emerge in the coming weeks as we receive the first data from the various clinical trials underway.
Clinical trials for remdesivir
Seven clinical trials have been initiated to determine whether remdesivir is a safe and effective treatment for COVID-19. Each of these was set up with unprecedented speed thanks to the remarkable efforts of the various groups involved, as well as the level of knowledge we had on remdesivir.
To some extent, the trials have had to be adaptive in design as our understanding of the disease itself continues to evolve. The virus emerged and spread at an intense speed and everyone is working quickly to understand it. Our interpretation of the results will also be shaped by what we continue to learn about the disease.
The order in which the trials were initiated mirrors the path of the pandemic. China initiated the first two studies in early February for patients with severe and moderate symptoms of the disease. Since then, an additional five trials have been initiated around the world.
Two Phase 3 studies are being run by Gilead in areas with a high prevalence of COVID-19 in the United States, Asia and Europe. One of these is for patients with severe disease and the other studies remdesivir in patients with more moderate symptoms. One of the many questions that these studies aim to answer is whether treatment duration can be shortened from 10 days to 5 days. The severe arm fully enrolled the number of patients it was originally designed for and we have now expanded the study so that thousands more patients can participate, including those on mechanical ventilation.
The U.S National Institute of Allergy and Infectious Disease (NIAID) began a global trial on February 21. This trial randomly assigns patients to treatment with either remdesivir or with a placebo to enable a controlled comparison of outcomes. The trial is enrolling approximately 800 patients with a broad spectrum of symptoms.
The World Health Organization is also conducting a global trial, Solidarity, and the Inserm DisCoVeRy trial recently began in Europe. A summary of remdesivir trials with upcoming data readouts can be found here.
We know that there is tremendous interest around when the data from these trials will be available and what they will tell us about remdesivir. We feel the urgency as we wait for the science to speak. With every day that goes by, the desperate need to equip healthcare workers and their patients with a safe, effective treatment becomes more pressing. We are working with intense speed to determine whether remdesivir could be an option and we are committed to sharing information when it becomes available to us.
We expect that we will have preliminary data from the study of remdesivir in severe patients at the end of April and will work quickly to interpret and share the findings. The publication of data from the China remdesivir trials rests with the Chinese investigators, but we have been informed that the study in patients with severe symptoms was stopped due to stalled enrollment. We look forward to reviewing the published data when available. In May, we anticipate the initial data from the placebo-controlled NIAID trial as well as data from the Gilead study of patients with moderate symptoms of COVID-19.
To a large extent, the timelines are determined by epidemiology and the numerous challenges that come with studying a treatment for a newly emerged disease. As with so much in this pandemic, this is unchartered territory for many of us involved in the process.
Ongoing collaboration
While it may feel like a long wait for data given the urgency of the situation, it has been only two months since the first clinical trials began. Given that it can take a year or more to have the first clinical data for an investigational treatment, it is remarkable that we expect to have the first remdesivir trial data so soon.
This speed is the result of strong collaboration and immense dedication among the many groups involved, from regulatory authorities to hospital administrators, clinicians and study investigators. As with all the work on remdesivir, everyone is driven by the same sense of urgency and a commitment to maintaining scientific rigor throughout.
All of us at Gilead are grateful to the many groups and organizations who are collaborating to find answers on remdesivir and above all, to the physicians and patients involved in the clinical trials. When we talk about trial results, we tend to think in terms of numbers, trends and statistics. We realize that behind each of these numbers is a patient who has agreed to take part in a trial and share the data from their personal experience. It is thanks to the thousands of patients like these and the physicians who are treating them, that we will be able to determine whether remdesivir can be used safely and effectively for many more patients in the future.
김대호 글로벌이코노믹 연구소장 tiger8280@g-enews.com